Abstract
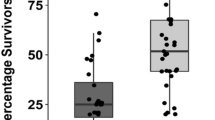
Genetically engineered crops that produce insecticidal toxins from Bacillus thuringiensis (Bt) are grown widely for pest control1. However, insect adaptation can reduce the toxins' efficacy2,3,4,5. The predominant strategy for delaying pest resistance to Bt crops requires refuges of non-Bt host plants to provide susceptible insects to mate with resistant insects2,3,4,5,6,7. Variable farmer compliance is one of the limitations of this approach. Here we report the benefits of an alternative strategy where sterile insects are released to mate with resistant insects and refuges are scarce or absent. Computer simulations show that this approach works in principle against pests with recessive or dominant inheritance of resistance. During a large-scale, four-year field deployment of this strategy in Arizona, resistance of pink bollworm (Pectinophora gossypiella) to Bt cotton did not increase. A multitactic eradication program that included the release of sterile moths reduced pink bollworm abundance by >99%, while eliminating insecticide sprays against this key invasive pest.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
James, C. Global status of commercialized biotech/GM crops: 2009. ISAAA Briefs 41 (ISAAA, Ithaca, New York, USA, 2009).
Kruger, M.J., Van Rensburg, J.B.J. & Van den Berg, J . Perspective on the development of stem borer resistance to Bt maize and refuge compliance at the Vaalharts irrigation scheme in South Africa. Crop Prot. 28, 684–689 (2009).
Tabashnik, B.E., Van Rensburg, J.B.J. & Carrière, Y. Field-evolved insect resistance to Bt crops: definition, theory, and data. J. Econ. Entomol. 102, 2011–2025 (2009).
Storer, N.P. et al. Discovery and characterization of field resistance to Bt maize: Spodoptera frugiperda (Lepidoptera: Noctuidae) in Puerto Rico. J. Econ. Entomol. 103, 1031–1038 (2010).
Bagla, P. Hardy cotton-munching pests are latest blow to GM crops. Science 327, 1439 (2010).
Gould, F. Sustainability of transgenic insecticidal cultivars: integrating pest genetics and ecology. Annu. Rev. Entomol. 43, 701–726 (1998).
Tabashnik, B.E., Gassmann, A.J., Crowder, D.W. & Carrière, Y. Insect resistance to Bt crops: evidence versus theory. Nat. Biotechnol. 26, 199–202 (2008).
Tabashnik, B.E., Dennehy, T.J. & Carrière, Y. Delayed resistance to transgenic cotton in pink bollworm. Proc. Natl. Acad. Sci. USA 102, 15,389–15,393 (2005).
Henneberry, T.J. & Naranjo, S.E. Integrated management approaches for pink bollworm in the southwestern United States. Integrated Pest Management Rev. 3, 31–52 (1998).
Naranjo, S.E. & Ellsworth, P.C. Fourteen years of Bt cotton advances IPM in Arizona. Southwest. Entomol. 35, 439–446 (2010).
Tabashnik, B.E. et al. Frequency of resistance to Bacillus thuringiensis in field populations of pink bollworm. Proc. Natl. Acad. Sci. USA 97, 12,980–12,984 (2000).
Tabashnik, B.E. et al. Association between resistance to Bt cotton and cadherin genotype in pink bollworm. J. Econ. Entomol. 98, 635–644 (2005).
Carrière, Y. et al. Cadherin-based resistance to Bt cotton in hybrid strains of pink bollworm: fitness costs and incomplete resistance. J. Econ. Entomol. 99, 1925–1935 (2006).
Dennehy, T.J. et al. Susceptibility of Southwestern Pink Bollworm to Bt toxins Cry1Ac and Cry2Ab2 in 2005. in Arizona Cotton Report (P-151) (Cooperative Extension, Agricultural Experiment Station, The University of Arizona, Tucson, USDA, 2007) <http://cals.arizona.edu/pubs/crops/az1437/az14373a.pdf>.
Liu, Y.B., Tabashnik, B.E., Dennehy, T.J., Patin, A.L. & Bartlett, A.C. Development time and resistance to Bt crops. Nature 400, 519 (1999).
Stone, G.D. Biotechnology and the political ecology of information in India. Hum. Organ. 63, 127–140 (2004).
Carrière, Y. et al. Long-term evaluation of compliance with refuge requirements for Bt cotton. Pest Manag. Sci. 61, 327–330 (2005).
Antilla, L. & Liesner, L. Program advances in the eradication of pink bollworm, Pectinophora gossypiella, in Arizona cotton. in Proceedings of the 2008 Beltwide Cotton Conferences, Nashville Tennessee, January 8–11, 2008, 1162–1168 (National Cotton Council of America, Memphis, Tennessee, USA, 2008).
Grefenstette, B., El-Lissy, O. & Staten, R.T. Pink Bollworm Eradication Plan in the US. (USDA, 2009). <http://www.aphis.usda.gov/plant_health/plant_pest_info/cotton_pests/downloads/pbw-erad-plan2-09.pdf>.
Gassmann, A.J., Carrière, Y. & Tabashnik, B.E. Fitness costs of insect resistance to Bacillus thuringiensis. Annu. Rev. Entomol. 54, 147–163 (2009).
Carrière, Y. et al. Long-term regional suppression of pink bollworm by Bacillus thuringiensis cotton. Proc. Natl. Acad. Sci. USA 100, 1519–1523 (2003).
Adamczyk, J.J. Jr. & Hubbard, D. Changes in populations of Heliothis virescens (F.) (Lepidoptera: Noctuidae) and Helicoverpa zea (Boddie) (Lepidoptera: Noctuidae) in the Mississippi Delta from 1986 to 2005 as indicated by adult male pheromone traps. J. Cotton Sci. 10, 155–160 (2006).
Micinski, S., Blouin, D.C., Waltman, W.F. & Cookson, C. Abundance of Helicoverpa zea and Heliothis virescens in pheromone traps during the past twenty years in northwestern Louisiana. Southwest. Entomol. 33, 139–149 (2008).
Storer, N.P., Dively, G.P. & Herman, R.A. Landscape effects of insect-resistant genetically modified crops. in Integration of Insect-Resistant Genetically Modified Crops within IPM Programs, (eds. Romeis, J., Shelton, A.M. & Kennedy, G.G.) 273–302 (Springer, The Netherlands, 2008).
Wu, K.M., Lu, Y.H., Feng, H.Q., Jiang, Y.Y. & Zhao, J.Z. Suppression of cotton bollworm in multiple crops in China in areas with Bt toxin-containing cotton. Science 321, 1676–1678 (2008).
Hutchison, W.D. et al. Areawide suppression of European corn borer with Bt maize reaps savings to non-Bt maize growers. Science 330, 222–225 (2010).
Naranjo, S.E. & Ellsworth, P.C. Fifty years of the integrated control concept: moving the model and implementation forward in Arizona. Pest Manag. Sci. 65, 1267–1286 (2009).
Ellsworth, P.C., Fournier, A. & Smith, T.D. Cotton Insect Losses (The University of Arizona, Cooperative Extension, 2010). <http://cals.arizona.edu/crops/cotton/insects/cil/cil.html>.
Anonymous. Pink Bollworm Eradication Current Status. (US Department of Agriculture, February, 2009). < http://www.aphis.usda.gov/plant_health/plant_pest_info/cotton_pests/downloads/PBW_prog_update2009.pdf>.
Krafsur, E.S. Sterile insect technique for suppressing and eradicating insect populations: 55 years and counting. J. Agric. Entomol. 15, 303–317 (1998).
Gould, F. & Schliekelman, P. Population genetics of autocidal control and strain replacement. Annu. Rev. Entomol. 49, 193–217 (2004).
Thomas, D.D., Donnelly, C.A., Wood, R.J. & Alphey, L.S. Insect population control using a dominant, repressible, lethal genetic system. Science 287, 2474–2476 (2000).
Alphey, N., Bonsall, M.B. & Alphey, L. Combining pest control and resistance management: synergy of engineered insects with Bt crops. J. Econ. Entomol. 102, 717–732 (2009).
U.S. Environmental Protection Agency. Scientific Advisory Panel, October 24–26, 2006: Evaluation of the resistance risks from using 100% Bollgard and Bollgard II cotton as part of a pink bollworm eradication program in the state of Arizona <http://www.epa.gov/scipoly/sap/meetings/2006/102406_mtg.htm#minutes>.
Sisterson, M.S., Antilla, L., Carrière, Y., Ellers-Kirk, C. & Tabashnik, B.E. Effects of insect population size on evolution of resistance to transgenic crops. J. Econ. Entomol. 97, 1413–1424 (2004).
Liu, Y.B. et al. Effects of Bt cotton and Cry1Ac toxin on survival and development of pink bollworm (Lepidoptera: Gelechiidae). J. Econ. Entomol. 94, 1237–1242 (2001).
Graham, H.M. & Mangum, C.L. Larval diets containing dye for tagging pink bollworm moths internally. J. Econ. Entomol. 64, 376–379 (1971).
Sims, M.A. et al. Arizona's multi-agency resistance management program for Bt cotton: sustaining the susceptibility of pink bollworm. in Proceedings of the 2001 Beltwide Cotton Conferences, January 9–13, 2001, Anaheim, California, 2, 1175–1179 (National Cotton Council of America, Memphis, Tennessee, USA, 2001).
Morin, S. et al. Three cadherin alleles associated with resistance to Bacillus thuringiensis in pink bollworm. Proc. Natl. Acad. Sci. USA 100, 5004–5009 (2003).
Morin, S. et al. DNA-based detection of Bt resistance alleles in pink bollworm. Insect Biochem. Mol. Biol. 34, 1225–1233 (2004).
Tabashnik, B.E. et al. DNA screening reveals pink bollworm resistance to Bt cotton remains rare after a decade of exposure. J. Econ. Entomol. 99, 1525–1530 (2006).
Tabashnik, B.E. et al. Asymmetrical cross-resistance between Bacillus thuringiensis toxins Cry1Ac and Cry2Ab in pink bollworm. Proc. Natl. Acad. Sci. USA 105, 11,889–11,894 (2009).
Tabashnik, B.E. et al. Dispersal of pink bollworm (Lepidoptera: Gelechiidae) males in transgenic cotton that produces a Bacillus thuringiensis toxin. J. Econ. Entomol. 92, 772–780 (1999).
Sokal, R.R. & Rohlf, F.J. Biometry (W.H. Freeman and Company, San Francisco, 1969).
SAS Institute. JMP 8.0, Cary, North Carolina, USA (2008).
Acknowledgements
We thank N. Alphey, D. Crowder, F. Gould, W. Hutchison, S. Naranjo, G. Rosenthal, R.L. Smith and K.M. Wu for their thoughtful comments and assistance; and E. Miller, D. Parker, M. Krueger, N. Manhardt and Arizona Cotton Research and Protection Council (ACRPC) staff members for their contributions to the eradication program. This work was supported by funding from the USDA-National Institute of Food and Agriculture program, ACRPC, Arizona Cotton Growers Association, Cotton Foundation, Cotton Inc., National Cotton Council, Western IPM Center, Arizona Pest Management Center, and Monsanto and Dow AgroSciences.
Author information
Authors and Affiliations
Contributions
M.S.S. conducted computer simulations; P.C.E. collected and summarized insecticide use data; L.A., L.L., M.W. and R.T.S. directed the eradication program and contributed to its design; J.A.F., G.C.U., A.J.Y., C.E.-K., and V.S.H. collected data; B.E.T., M.S.S., P.C.E., T. J. D., L.A., R.T.S., J.A.F., G.C.U., X. L. and Y.C. contributed to research design; Y.C. analyzed data. B.E.T. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
Research reported in this article was supported by organizations that may gain or lose financially through its publication: Arizona Cotton Growers Association, Arizona Cotton Research and Protection Council (ACRPC), Cotton Foundation, Cotton Incorporated, National Cotton Council, Monsanto and Dow AgroSciences. L.A. and R.T.S. are independent contractors paid by ACRPC. L.L. and M.W. are employees of ACRPC. T.J.D. is an employee and stockholder of Monsanto Company. B.E.T. is a coauthor of a patent application on engineering modified Bt toxins to counter pest resistance, which is related to research described by Soberon et al. (Science 318, 1640–1642 (2007)).
Supplementary information
Supplementary Text and Figures
Supplementary Table 1, Supplementary Methods and Supplementary Figs. 1,2 (PDF 164 kb)
Rights and permissions
About this article
Cite this article
Tabashnik, B., Sisterson, M., Ellsworth, P. et al. Suppressing resistance to Bt cotton with sterile insect releases. Nat Biotechnol 28, 1304–1307 (2010). https://doi.org/10.1038/nbt.1704
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1704
This article is cited by
-
The evaluation of resistance risk to Cry2Ab and cross-resistance to other insecticides in Helicoverpa armigera
Journal of Pest Science (2024)
-
Impact of simultaneous treatment of gamma irradiation and Bacillus thuringiensis on cotton leaf worm Spodoptera littoralis (Boisd.) (Noctuidae: Lepidoptera)
Egyptian Journal of Biological Pest Control (2022)
-
Ectopic expression of Xenorhabdus nematophila chitinase in tobacco confers resistance against Helicoverpa armigera
Plant Cell, Tissue and Organ Culture (PCTOC) (2022)
-
A high-efficiency Agrobacterium-mediated transient expression system in the leaves of Artemisia annua L.
Plant Methods (2021)
-
Integrated pest management: good intentions, hard realities. A review
Agronomy for Sustainable Development (2021)